
The atomic radius is a term used to describe the size of the atom. The radii of neutral atoms range from 30 to 300 pm or trillionths of a meter. 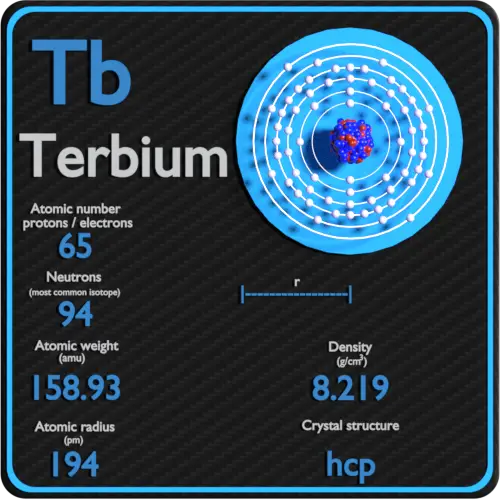
In practice, the value is obtained by measuring the diameter of an atom and dividing it in half. IMPORTANT: You must include your IP address, otherwise we won't know which address to unblock! You can check your actual IP address (as seen by the outside world) using a free service such as Ĭopyright © 2022 CrystalMaker Software Ltd. The atomic radius is the distance from the atomic nucleus to the outermost stable electron of a neutral atom. In the case of Carbon There are cool facts about Carbon that most dont know about. Please contact us to request that we unblock your access. Note: Learn more about the atomic radius here. Explanation: The distance from the center of the nucleus to the outermost shell of the electron is known as the atomic radius of an element. Have we made a mistake? If you believe we have mistakenly blocked your access, we apologise. Answer: The elements in increasing order of atomic radius: oxygen, carbon, aluminum, potassium. You, or another user with the same IP address, has been detected posting spam, attempting to hack this site, or making a denial-of-service attempt. So please purchase the appropriate licence(s) from us, and delete any stolen software from your computer. Without this income there would be no software. We rely on software sales to fund our research and development. You, or another user with the same IP address, has been detected using an illegal, stolen ("hacked") copy of our software.ĭo not use stolen or "hacked"/"cracked" software: it's illegal - and it's seriously uncool: nobody likes a cheat. Access Denied You are forbidden to access this website because: Either:
0 Comments
Leave a Reply. |
 RSS Feed
RSS Feed
